Retrospective analysis of para-aortic lymph node recurrence in locally advanced cervix cancers treated with “pelvic-only field” radiotherapy
Analyse rétrospective de la rechute ganglionnaire para-aortique dans les cancers du col utérin localement avancés traités par radiothérapie « champ pelvien seul »
EN. Séka1,2, KD. N’chiépo1, GG. N’da1, BAW. Mossé1, S. El Majjaoui1,3, N Benjaafar1,3.
1 Department of radiotherapy of National Institute of Oncology of Rabat, Morocco.
2 Department of hematology/immunology and oncology of faculty of medicine of Felix Houphouet Boigny, University of Abidjan, Ivory Coast.
3 Department of Radiotherapy of faculty of medicine and pharmacy of Mohammed V University of Rabat, Morocco.
DOI: 10.54266/ajo.2.1.9.MXhLpWr8cI
ABSTRACT
INTRODUCTION: Previous studies suggested that treating para-aortic lymph node micro-metastases with extended-field radiotherapy could result in higher rate of tumor control in high-risk locally advanced cervical cancers. Our study aimed to assess the patterns of recurrences following pelvic-only irradiation, with an emphasis on recurrence in para-aortic lymph node. MATERIALS AND METHODS: We retrospectively evaluated pattern of recurrences in patients with cervical cancers who were treated with definitive pelvic radiotherapy at our institution from 2013 to 2016. Univariate and multivariate analyses were performed to identify risk factors associated with para-aortic lymph node recurrence. RESULTS: We enrolled 463 patients in the study. The median of follow-up was 63.5 months. Five-years overall survival, recurrence-free survival, and para-aortic lymph node recurrence rates were respectively 78.9%, 76.2% and 6.5%. During follow-up, 23.33% experienced recurrences. Most often tumor recurred locally (42.60%). Para-aortic recurrences were found in 26 patients. Patients’ age (≤57 years) (HR=4.9, p=0.002), histological subtype (adenocarcinoma) (HR=2.7, p=0.035), presence of pelvic lymph node metastasis (HR=0.3, p=0.037) and absence of brachytherapy (HR=6.5, p<0.0001) were significantly associated with para-aortic lymph node (PALN) recurrence in multivariate analysis. CONCLUSION: Recurrences following pelvic-only irradiation were frequently observed in the pelvis. Clinical characteristics associated with a high risk of PALN recurrence were young age, absence of brachytherapy, presence of pelvic lymph node metastases, and the adenocarcinoma subtype. However, further studies with a greater number of patients need to be undertaken.
KEYWORDS: Radiotherapy; Recurrences; Para-aortic; Cervical cancer.
RESUME
INTRODUCTION : Les études antérieures ont suggéré que le traitement des micro-métastases des aires ganglionnaires para-aortiques par radiothérapie à champ étendu pourrait résulter en un taux de contrôle tumoral plus élevé dans les cancers utérins localement avancés de haut risque. Notre étude avait pour but d’évaluer les modes de récidives après irradiation pelvienne seule, tout en mettant l’accent sur les récidives ganglionnaires para-aortiques. MATERIELS ET METHODES : Nous avons évalué d’une façon rétrospective les modalités de récidive chez les patientes atteintes de cancers du col utérin qui ont été traitées par radiothérapie exclusive dans notre établissement de 2013 à 2016. Des analyses univariées et multivariées ont été réalisées pour identifier les facteurs de risque associés à la rechute ganglionnaire para-aortique. RESULTATS : Nous avons inclus 463 patientes dans l’étude. La médiane de suivi était de 63,5 mois. Les taux de survie globale, de survie sans récidive et de rechute ganglionnaire para-aortique à cinq ans étaient respectivement de 78,9%, 76,2% et 6,5%. Au cours du suivi, 23,33% ont présenté des récidives. Les récidives étaient le plus souvent locales (42,60%). Des récidives para-aortiques ont été retrouvées chez 26 patients. L’âge des patients (≤57 ans) (HR=4,9, p=0,002), le sous-type histologique (adénocarcinome) (HR=2,7, p=0,035), la présence de métastases ganglionnaires pelviennes (HR=0,3, p=0,037) et l’absence de curiethérapie (HR=6,5, p<0,0001) étaient significativement associés à la récidive ganglionnaire para-aortique en analyse multivariée. CONCLUSION : Les récidives après irradiation pelvienne seule, étaient fréquemment observées dans le pelvis. Les caractéristiques cliniques associées à un risque élevé de récidive ganglionnaire para-aortique étaient le jeune âge, l’absence de curiethérapie, la présence de métastase ganglionnaire pelvienne et le sous-type adénocarcinome. Cependant, d’autres études avec un plus grand nombre de patientes doivent être entreprises.
MOTS-CLES : Radiothérapie ; Récidives ; Para-aortique ; Cancer col utérin.
INTRODUCTION
Radiotherapy is efficacious, and constitutes the cornerstone in managing locally advanced carcinomas of the cervix. Despite its effectiveness on long-term tumor control, recurrence is often observed during the follow-up period (1). Most of these recurrences occur within the first two to three years following treatment completion and are located in the pelvis, either locally or regionally (pelvic lymph nodes) (2,3). Para-aortic lymph nodes (PALN) are the second level of lymphatic drainage in cervical cancer. They are also frequently involved in recurrence and play an important role in metastatic dissemination. In locally advanced tumors, microscopic metastases can be relatively frequent in PALN. Imaging techniques currently available for primary assessment can miss these microscopic metastases that could be responsible for tumor recurrence (4). Theoretically, treating occult microscopic PALN metastases with prophylactic extended-field radiation would translate into reduction of PALN and then systemic recurrences. However, the efficacy of this prophylactic extended-field approach has not been proved (5). In previous studies, some prognosis factors have been identified to be correlated with PALN recurrence. These are extensive parametrial involvement, pelvic lymph node metastases, and high serum level of SCC (6). Reports on the efficiency of prophylactic extended-field on the basis of these factors are contradictory and do not provide clear guidance (5). In our institution, patients without overt PALN metastasis are treated with pelvic-only radiation field irrespective of parametria or pelvic wall involvement and pelvic node status. Thus, we conducted this study to assess the patterns of recurrence following pelvic-only irradiation with an emphasis on recurrence in PALN.
MATERIELS ET METHODES
Patients’ selection
We carried out a retrospective selection of patients with cervical cancer treated with radiotherapy at the National Institute of Oncology of Rabat (Morocco) from 2013 to 2016.
Patients with the following characteristics were included: histologically confirmed carcinoma of the uterine cervix, clinical examination with a report on primary tumor extension to the vagina, parametria, and pelvic wall, tumor extension assessment with pelvic MRI or CT-scan and chest-abdomen CT-scan, treatment completion with a total dose of at least 66 Gy (EQD2Gy), followed-up for at least six months after treatment completion, imaging and pathological substantiation of the recurrence. We did not include patients with the following: primary metastatic cancer, primary para-aortic lymph node enlargement (on imaging), patients with a second cancer, progression before treatment completion. Dosage of SCC (squamous cell carcinoma) tumor marker was not mandatory.
Treatment description
The treatment plan used 3-Dimensional Conformal Radiation Therapy (3DCRT) technique for all cases. Radiotherapy was delivered following two successive steps that were external Radiation Therapy course (EBRT) and brachytherapy course or an EBRT boost if brachytherapy was not feasible. Pelvic Radiation Therapy (PRT) delivered a total dose of 45 or 46 Gy in conventional fractionation (1.8 or two Gy/fraction per day and five fractions per week). For patients with pelvic node or pelvic wall invasion, boost doses of 10 Gy to 14 Gy and nine Gy to 10 Gy were given to the node(s) or parametria respectively. Patients received concurrent chemotherapy during this step. Brachytherapy was giving at a high dose rate (HDR). The EBRT field covered the gross primary tumor, the whole genital tract (uterine cervix, uterine corpus, vaginal portion, parametria, and adnexa), and the regional pelvic nodes (including the common, internal and external iliac, obturator and presacral lymph nodes). For the EBRT boost field, the pelvis node region was excluded from the pelvic field. Treatment was delivered in conventional fractionation with a linear accelerator. Concurrent chemotherapy regimen was intravenous infusion of cisplatin 40 mg/m² (maximal dose=70 mg) once a week during the PRT course.
Follow-up
Patients were examined every three or four months during the first two years after treatment completion, then every six months for the three subsequent years, then annually. Treatment response, disease recurrence, and complications were recorded. Tumor response was assessed using clinical examination. Imaging was not performed systematically. An MRI or CT-scan was required for patients who were symptomatic. All recurrences were histologically confirmed if the site was accessible for biopsy.
Statistical analysis
Lymph node diagnosis was based on imaging findings (CT-scan or MRI). A lymph node was considered metastatic if it had the following characteristics: short-axis superior or equal to 10 mm, or rounded with central necrosis. Pelvic wall involvement was diagnosed on gynecological examination and/or with pelvic CT-scan or MRI. Only the first site of recurrence was considered in this study. Recurrences were grouped into three categories according to the natural history of cervical carcinomas: the first group was those located in the pelvic (local recurrence and/or pelvic lymph node metastasis), the second group was para-aortic lymph node metastasis with or without first group, involvement and the third group was distant metastasis with or without first or second group involvement. Overall survival, progression-free survival, and pelvic, para-aortic, and distant failure rates were evaluated with the Kaplan-Meier method. We used the log-rank method for univariate analysis, and variables that showed a trend (p<0.10) underwent a multivariate analysis. The Cox regression model was developed using forward stepwise regression. Results were considered significant for p-value less than 0.05. These analyses were performed with SPSS data analysis software.
RESULTS
Four hundred sixty-three patients were included in this study. The average age was 57 years and approximately 56% were less than 57 years old. Pathologically, tumors were overwhelmingly squamous cell subtype (90.6%). Tumor sizes were equal or superior to four cm in 73% of patients. According to the FIGO staging system, tumors were classified as stage IB (40 patients: 8.7%), stage IIA (46 patients: 9.9%), stage IIB (115 patients: 24.8%), stage IIIA (12 patients: 2.6%), stage IIIB (76 patients: 16.4%) and stage IVA (36 patients: 7.78%). One hundred thirty-six (29.37%) patients had tumors that invaded the pelvic wall (extensive parametria invasion). Pelvic lymph node metastases were found in 146 patients (31.53%) and most patients had hemoglobin levels above 10 g/mm3. Concurrent chemotherapy and brachytherapy were performed in 92.87% and 83.37% respectively (Table I).
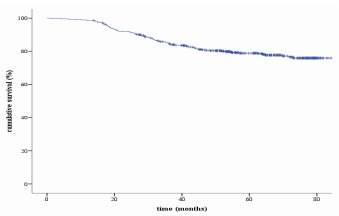
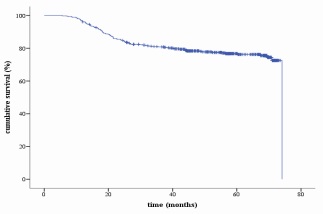
The median of follow-up was 63.5 months (ranging from 1.2 to 109.23 months). The median time to recurrence was 20.33 months (ranging from 5.6 to 74.1 months). Five-years overall survival (OS), recurrence-free survival (RFS), and para-aortic lymph node recurrence (PALNR) rates were respectively 78.9%, 76.2% and 6.5% (Figures 1 to 3).
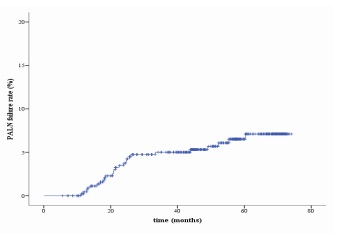
During follow-up, 108 patients (23.33%) experienced recurrences. Among them, 55 (60.19%) were isolated recurrences and 43 (39.81%) occurred in more than one anatomical site. Para-aortic recurrences were found in 26 patients and were distributed as follows: isolate para-aortic recurrence (Two patients), para-aortic with pelvis recurrences (Nine patients), para-aortic with metastatic recurrences (Eight patients), and para-aortic with pelvic and metastatic recurrences (Seven patients) (Table II).
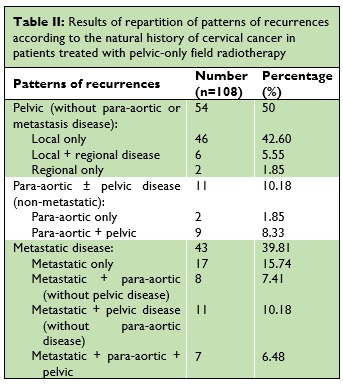
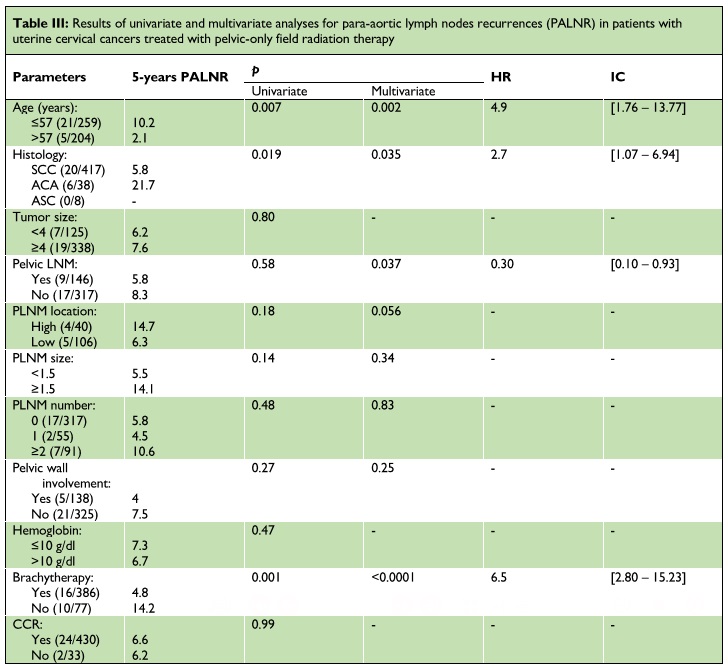
Patients’ age (inferior or equal to 57 years), histological subtype (adenocarcinoma), and absence of brachytherapy were significantly associated with PALNR in both univariate and multivariate analysis. The presence of pelvic lymph node metastasis did not show significance in univariate analysis. However, it was correlated with PALNR in multivariate analysis (Table III).
DISCUSSION
Our study aimed at patterns of recurrences in uterine cervical carcinomas with an emphasis on para-aortic lymph node recurrences. All of our patients were treated with pelvic-only radiation therapy. Five-years overall and recurrence-free survival of patients in our study (respectively 78.9% and 76.2%) was comparable to data in the literature (7–9). At a median follow-up of 63.5 months, approximately 23% of patients showed recurrences. Most of these recurrences occurred locally (50%). In literature, few studies have focused on patterns of recurrences. Vale and al (10), in a meta-analysis of 13 randomized trials, found that 35% of their patients experienced treatment failure. Of these, 45% were in the pelvis alone and 34% were distant failure alone. In the retroEMBRACE study, distant recurrence was more frequent than local ones. Thirty percent of patients had recurrences. Twenty-one percent of these patients experienced local recurrence, and 57% had distant lesions (11). In our study, a non-negligible number of patients did not receive brachytherapy (16.63%) which certainly led to the high rate of local recurrences. The absence of brachytherapy has been proved an adverse prognosis factor (12) that leads to significant rates of local recurrence. Other tumor characteristics like large tumor size, advanced FIGO stage have probably influenced our results. As found in other studies (13,14), only 26 (5.6%) patients presented para-aortic lymph node recurrence. Among them, only two patients had an isolated para-aortic recurrence. Most of the recurrences occurred concurrently with local recurrence. These results show that pelvic only irradiation in locally advanced cancer of the uterine cervix did not lead to more frequent para-aortic recurrence overall. However, it is important to consider metastatic recurrence as para-aortic lymph nodes are implicated in the process of metastatic dissemination.
Overall, para-aortic lymph node recurrence rate was 6.5% at five years. Factors associated with para-aortic recurrence in our study were young age (less or equal to 57 years), absence of brachytherapy, presence of metastatic pelvic lymph nodes, and adenocarcinoma subtype. However, the absence of brachytherapy is not a specific factor of para-aortic failure, as it constitutes an adverse factor for overall treatment outcomes. Some authors have already investigated the concern of para-aortic lymph node recurrence in locally advanced cervical cancers treated with radiotherapy. Huang et al (6) found that SSC level, pelvic wall involvement, and presence of pelvic lymph node metastasis were associated with para-aortic relapse. However, in this study, only squamous cell carcinomas were included and parameters explored for the concern of pelvic lymph nodes metastases were either the presence or the absence of pelvic lymph nodes. The authors did not explore lymph node characteristics such as location, size, or number. Wang and al (14) addressed them in their study and they found that besides the pelvic wall involvement, the number of involved nodes (more or equal than two) in the pelvic was associated with para-aortic recurrence. The location of metastasis in the common iliac lymph nodes did not show significance in multivariate analysis. In our study, we also addressed these characteristics in pelvic lymph nodes. We did not find an association between these characteristics and para-aortic recurrence. However, the location of lymph nodes had a borderline p-value (0.056). Unlike the finding of the precedent two studies, pelvic wall involvement did not correlate with para-aortic recurrence in our study. The adenocarcinoma subtype was associated with para-aortic recurrence in our study. Note that the aggressiveness of adenocarcinomas compared to that of squamous cell carcinomas is subject to controversy. Some studies have shown worse prognostic of adenocarcinomas and others not. Hu and al (15) compared the treatment outcomes between squamous cell carcinoma and adenocarcinoma of the cervix. They showed that adenocarcinomas were more aggressive, and had more para-aortic metastatic lymph nodes than squamous cell carcinoma. Three-year disease-free survival, overall survival, local control rates, and distant control rates were worse for adenocarcinoma compared to SCC. In a propensity score matching study, Yin and al (16) had also demonstrated that adenocarcinomas had worse treatment outcomes compared to the squamous cell subtype. In addition, in a retrospective study based on medical and histopathological record review, Chen and al (13) found that patients with adenocarcinomas had worse PFS, distant metastasis-free survival, and OS than patients with SCC. Katanyoo and al (17) did not show any difference in the pattern of recurrence between the two subtypes in their study. Rose and al (18), also, demonstrated similar survival and outcomes between adenocarcinomas and SCC when treated with concurrent chemo-radiation. Our study had some limitations. Retrospective analysis of data from a single institution could have an impact on the quality of our results. The tumor marker SCC was not considered among risks factors, as its measurement is not mandatory in our center. Furthermore, SCC is not a marker for the other histological subtype. In addition, the percentage of patients that had para-aortic relapse were small. Only two patients had isolate PALN recurrences. The small number of PALN recurrence had certainly influenced our results.
CONCLUSION
Our study demonstrated that pelvic-only radiotherapy with concurrent chemotherapy did not lead to significant PALN recurrence in cervical cancer. Recurrence was observed mostly in the pelvis. Clinical characteristics that correlated to PALN recurrence were young age, absence of brachytherapy, presence of pelvic lymph node metastasis, and the adenocarcinoma subtype. However, the location of lymph nodes in the pelvic must be taken into consideration. Further studies with a greater number of patients or multi-institutional must be undertaken.
CONFLICTS OF INTEREST: The authors have declared no conflict of interest.
STATEMENTS
We thank the International Atomic Energy Agency (IAEA) for funding for our radiation therapy specialist training course.
REFERENCES
- de Foucher T, Bendifallah S, Ouldamer L, Bricou A, Lavoue V, Varinot J, et al. Patterns of recurrence and prognosis in locally advanced FIGO stage IB2 to IIB cervical cancer: Retrospective multicentre study from the FRANCOGYN group. European Journal of Surgical Oncology. 2019 Apr;45(4):659–65. DOI : 10.1016/j.ejso.2018.11.014
- Bhatla N, Aoki D, Sharma DN, Sankaranarayanan R. Cancer of the cervix uteri. Int J Gynecol Obstet. 2018 Oct;143:22–36. DOI : 10.1002/ijgo.12611
- Algarve University Hospital Center, Rua Leão Penedo, Faro, Portugal, Linhares Moreira AS, Cunha TM, Portuguese Oncology Institute of Lisbon Francisco Gentil, Lisbon, Portugal, Esteves S, Portuguese Oncology Institute of Lisbon Francisco Gentil, Lisbon, Portugal. Cervical cancer recurrence – can we predict the type of recurrence? Diagn Interv Radiol. 2020 Sep 8;26(5):403–10. DOI : 10.5152/dir.2020.19437
- Gouy S, Morice P, Narducci F, Uzan C, Gilmore J, Kolesnikov-Gauthier H, et al. Nodal-staging surgery for locally advanced cervical cancer in the era of PET. The Lancet Oncology. 2012 May;13(5):e212–20. DOI : 10.1016/S1470-2045(12)70011-6
- Wang W, Zhou Y, Wang D, Hu K, Zhang F. Prophylactic Extended-Field Irradiation in Patients With Cervical Cancer: A Literature Review. Front Oncol. 2020 Oct 2;10:579410. DOI : 10.3389/fonc.2020.579410
- Huang E-Y, Wang C-J, Chen H-C, Fang F-M, Huang Y-J, Wang C-Y, et al. Multivariate Analysis of Para-Aortic Lymph Node Recurrence After Definitive Radiotherapy for Stage IB-IVA Squamous Cell Carcinoma of Uterine Cervix. International Journal of Radiation OncologyBiologyPhysics. 2008 Nov;72(3):834–42. DOI : 10.1016/j.ijrobp.2008.01.035
- Eifel PJ, Winter K, Morris M, Levenback C, Grigsby PW, Cooper J, et al. Pelvic Irradiation With Concurrent Chemotherapy Versus Pelvic and Para-Aortic Irradiation for High-Risk Cervical Cancer: An Update of Radiation Therapy Oncology Group Trial (RTOG) 90-01. JCO. 2004 Mar 1;22(5):872–80. DOI : 10.1200/JCO.2004.07.197
- Parker K, Gallop-Evans E, Hanna L, Adams M. Five Years’ Experience Treating Locally Advanced Cervical Cancer With Concurrent Chemoradiotherapy and High-Dose-Rate Brachytherapy: Results From a Single Institution. International Journal of Radiation OncologyBiologyPhysics. 2009 May;74(1):140–6. DOI : 10.1016/j.ijrobp.2008.06.1920
- Giavedoni ME, Staringer L, Garrido R, Bertoncini C, Sardi M, Perrotta M. Experience with concurrent chemoradiotherapy treatment in advanced cervical cancer: results from a hospital in Argentina. ecancer [Internet]. 2019 Apr 2 [cited 2022 Mar 16];13. Available from: https://ecancer.org/journal/13/full/919-experience-with-concurrent-chemoradiotherapy-treatment-in-advanced-cervical-cancer-results-from-a-hospital-in-argentina.php. DOI : 10.3332/ecancer.2019.919
- Reducing Uncertainties About the Effects of Chemoradiotherapy for Cervical Cancer: A Systematic Review and Meta-Analysis of Individual Patient Data From 18 Randomized Trials. JCO. 2008 Dec 10;26(35):5802–12. DOI : 10.1200/JCO.2008.16.4368
- Tan L-T, Pötter R, Sturdza A, Fokdal L, Haie-Meder C, Schmid M, et al. Change in Patterns of Failure After Image-Guided Brachytherapy for Cervical Cancer: Analysis From the RetroEMBRACE Study. International Journal of Radiation OncologyBiologyPhysics. 2019 Jul;104(4):895–902. DOI : 10.1016/j.ijrobp.2019.03.038
- Han K, Milosevic M, Fyles A, Pintilie M, Viswanathan AN. Trends in the Utilization of Brachytherapy in Cervical Cancer in the United States. International Journal of Radiation OncologyBiologyPhysics. 2013 Sep;87(1):111–9. DOI : 10.1016/j.ijrobp.2013.05.033
- Chen C-S, Ou Y-C, Lin H, Wang C-J, Chen H-C, Fang F-M, et al. Analysis of prognostic factors and clinical outcomes in uterine cervical carcinoma with isolated para-aortic lymph node recurrence. Am J Transl Res. 2019;11(12):7492–502.
- Wang W, Wang D, Liu X, Zhou Y, Ma J, Hou X, et al. Risk factors associated with Para-Aortic Lymph Node Failure after pelvic irradiation in patients with Cervical Cancer. J Cancer. 2020;11(17):5099–105. DOI : 10.7150/jca.45520
- Hu K, Wang W, Liu X, Meng Q, Zhang F. Comparison of treatment outcomes between squamous cell carcinoma and adenocarcinoma of cervix after definitive radiotherapy or concurrent chemoradiotherapy. Radiat Oncol. 2018 Dec;13(1):249. DOI : 10.1186/s13014-018-1197-5
- Yin K-C, Lu C-H, Lin J-C, Hsu C-Y, Wang L. Treatment outcomes of locally advanced cervical cancer by histopathological types in a single institution: A propensity score matching study. Journal of the Formosan Medical Association. 2018 Oct;117(10):922–31. DOI : 10.1016/j.jfma.2018.07.002
- Katanyoo K, Sanguanrungsirikul S, Manusirivithaya S. Comparison of treatment outcomes between squamous cell carcinoma and adenocarcinoma in locally advanced cervical cancer. Gynecologic Oncology. 2012 May;125(2):292–6. DOI : 10.1016/j.ygyno.2012.01.034
- Rose PG, Java JJ, Whitney CW, Stehman FB, Lanciano R, Thomas GM. Locally advanced adenocarcinoma and adenosquamous carcinomas of the cervix compared to squamous cell carcinomas of the cervix in Gynecologic Oncology Group trials of cisplatin-based chemoradiation. Gynecologic Oncology. 2014 Nov;135(2):208–12. DOI : 10.1016/j.ygyno.2014.08.018